Executive Summary
– Soil acidification is a natural process in high rainfall environments where leaching slowly acidifies soil over time.
– Intensive agriculture can speed up soil acidification through many processes – increasing leaching, addition of fertilizers, removal of produce and build-up of soil organic matter.
– Of all the major fertilizer nutrients, nitrogen is the main nutrient affecting soil pH, and soils can become more acidic or more alkaline depending on the type of nitrogen fertilizer used.
– Nitrate-based products are the least acidifying of the nitrogen fertilizers, while ammonium-based products have the greatest potential to acidify soil.
– Soil acidification due to use of phosphorus fertilizers is small compared to that attributed to nitrogen, due to the lower amounts of this nutrient used and the lower acidification per kg phosphorus. Phosphoric acid is the most acidifying phosphorus fertilizer.
– Potassium fertilizers have little or no effect on soil pH.
Background
Soil acidification is a widespread natural phenomenon in regions with medium-to-high rainfall. Agricultural production systems can accelerate soil acidification processes through:
– Perturbation of the natural cycles of nitrogen (N), phosphorus (P) and sulfur (S) in soil
– Removal of agricultural produce from the land
– The addition of fertilizers and soil amendments that can either acidify soil or make it more alkaline
Source: Kennedy 1986
Changes in soil pH may be advantageous or detrimental depending on the starting pH of the soil and the direction and speed of pH change – for example decreases in soil pH in alkaline soils may be advantageous for crop production due to benefits in terms of the availability of P and micronutrients e.g. zinc (Zn) (Mitchell et al. 1952).
On the other hand, decreases in soil pH for a highly acidic soil may be detrimental in terms of increasing crop susceptibility to toxicity induced by increased solubility of aluminium (Al) or manganese (Mn) as soil pH falls (Wright 1989).
Key processes and reasons for changes in soil pH in agricultural systems are as follows.
Fertilizer use
Use of mineral or organic fertilizers in agriculture increases inputs of nutrients to soils, and the form in which the nutrients are applied and their fate in the soil-plant system determine the overall effects on soil pH.
Macronutrients (N, P, potassium (K) and S) have the major effects on soil pH as they are added in much larger quantities to soil than micronutrients.
Nitrogen
The form of N and the fate of N in the soil-plant system is probably the major driver of changes in soil pH in agricultural systems.
Nitrogen can be added to soils in many forms, but the predominant forms of fertilizer N used are:
– Urea (CO(NH₂)₂)
– Monoammonium phosphate (NH₄H₂PO₄)
– Diammonium phosphate ((NH₄)₂HPO₄)
– Ammonium nitrate (NH₄NO₃)
– Calcium ammonium nitrate (CaCO₃+NH₄(NO₃))
– Ammonium sulfate ((NH₄)₂SO₄)
– Urea ammonium nitrate (a mixture of urea and ammonium nitrate)
– Ammonium polyphosphate ([NH₄PO₃]n)
The key molecules of N in terms of changes in soil pH are the uncharged urea molecule ([CO(NH₂)₂]0), the cation ammonium (NH₄+) and the anion nitrate (NO₃-). The conversion of N from one form to the other involves the generation or consumption of acidity, and the uptake of urea, ammonium or nitrate by plants will also affect acidity of soil (Figure 1).
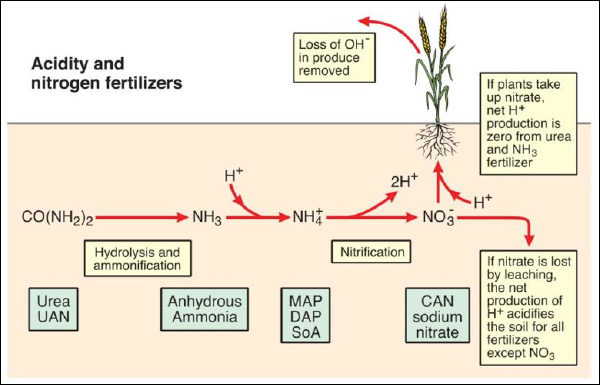
Figure 1. Soil acidity and nitrogen fertilizers (modified from (Davidson 1987)). MAP = monoammonium phosphate, DAP = diammonium phosphate, SoA = sulfate of ammonia, CAN = calcium ammonium nitrate, sodium nitrate
It can be seen in Figure 1 that ammonium-based fertilizers will acidify soil as they generate two H⁺ ions for each ammonium molecule nitrified to nitrate. The extent of acidification depends on whether the nitrate produced from ammonium is leached or is taken up by plants.
If nitrate is taken up by plants the net acidification per molecule of ammonium is halved compared to the scenario when nitrate is leached. This is due to the consumption of one H⁺ ion (or excretion of OH⁻) for each molecule of nitrate taken up – this is often observed as pH increases in the rhizosphere (Smiley and Cook 1973).
Anhydrous ammonia and urea have a lower acidification potential compared to ammonium-based products as one H⁺ ion is consumed in the conversion to ammonium. Nitrate-based fertilizers have no acidification potential and actually can increase soil pH as one H⁺ ion is absorbed by the plant (or OH⁻ excreted) in the uptake of nitrate.
Phosphorus
The form of phosphorus fertilizer added to soil can affect soil acidity, principally through the release or gain of H⁺ ions by the phosphate molecule depending on soil pH (Figure 2).
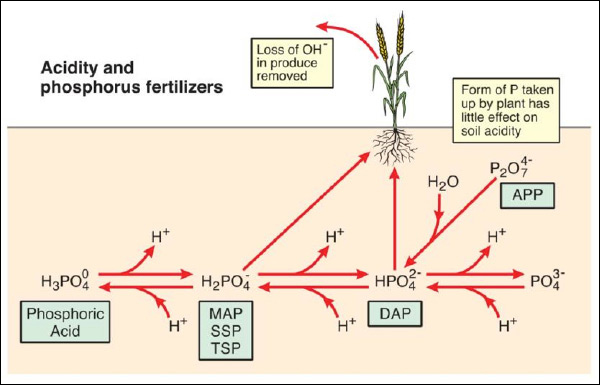
Figure 2. Soil acidity and P fertilizers. MAP = monoammonium phosphate, DAP = diammonium phosphate,
SSP = single superphosphate, TSP = triple superphosphate, APP = ammonium polyphosphate.
If phosphoric acid (PA) is added to soil, the molecule will always acidify soil as H⁺ ions will be released – one H⁺ ion if the soil pH is less than ~6.2 and two H⁺ ions is the soil pH is above 8.2.
Monoammonium phosphate (MAP), single superphosphate (SSP) and triple superphosphate (TSP) all add P to soil in the form of the H₂PO₄⁻ ion, which can acidify soil with a pH greater than 7.2 but has no effect on soil pH in acidic soils. The form of P in diammonium phosphate (DAP) is HPO₄²⁻ which can make acidic soils (pH<7.2) more alkaline but has no effect on soil with a pH>7.2. The hydrolysis of ammonium polyphosphate (APP), where the P present as the P₂O₇⁴⁻molecule converts to HPO₄²⁻, is pH neutral and hence any acidification due to adding P can be regarded as similar to DAP. SSP or TSP are sometimes declared to cause soil acidification due to reaction products being very acidic;
Ca(H₂PO₄)₂+ ₂H₂O -> CaHPO₄ + H⁺ + H₂PO₄⁻
but in soils with pH values less than 7.7 the following reaction neutralizes the acidity produced so that there is no net acidification;
CaHPO₄ + H₂O -> Ca₂+ + H₂PO₄⁻ + OH⁻
In high pH soils (pH >7.2), dissociation of H+ ion from the H₂PO₄⁻ molecule will generate some acidity.
Crop uptake of P has little effect on soil acidity due to the small amounts of fertilizer P taken up in any one year – hence fertilizer chemistry dominates pH changes and significant differences in rhizosphere pH have not been observed for uptake of different orthophosphate ions.
Sulfur
The form of sulfur fertilizer added to soil can affect soil acidity, principally through the release of H⁺ ions by the addition of elemental S (S⁰) or thiosulfate (S₂O3²⁻, in ammonium thiosulfate – ATS) (Figure 3). However, the amounts of S added to soil and taken up by plants are generally small in comparison to N.
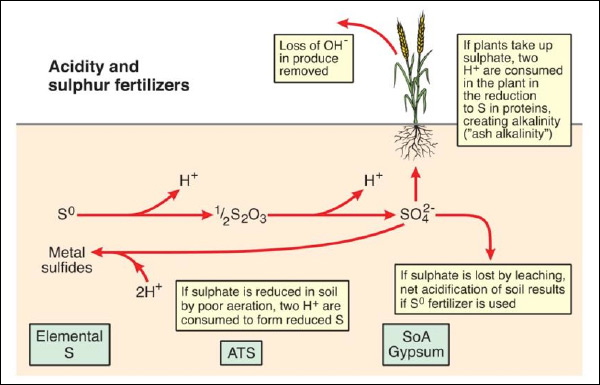
Figure 3. Soil acidity and S fertilizers. S⁰ = elemental S, ATS = ammonium thiosulfate, SoA = sulfate of ammonia.
For each molecule of S⁰ added to soil, two H⁺ ions will be generated, and these can be balanced through plant uptake by either uptake of H⁺ (same as excretion of OH⁻ ions) or the generation of OH⁻ (effectively organic anions) within the plant to form alkaline plant material (“ash alkalinity”). Where produce is removed (which is often the case in agricultural systems) net acidification of soil will occur if S⁰ or ATS are used.
Potassium
The form in which potassium is added to soil – either muriate of potash (KCl) or sulfate of potash (K₂SO₄) – has no effect on soil acidification.