Despite phosphorus (P) availability from geologic deposits distributed around the globe, widespread P deficiency in soils limits the growth and productivity of plants in many parts of the world. Because of this, growers commonly add this nutrient to their fields to improve crop yield and quality. Polyphosphate is an excellent liquid P fertilizer used to increase agricultural production.
Production
Phosphoric acid is the starting material for most commercial phosphate fertilizers. However, its acidity and some of its chemical properties make this material difficult to apply directly.
The most common ammonium polyphosphate fertilizers have a N-P₂O₅-K₂O (nitrogen, phosphorus and potassium) composition of 10-34-0 or 11-37-0. Polyphosphate fertilizers offer the advantage of a high nutrient content in a clear, crystal-free fluid that remains stable within a wide temperature range and stores well for long periods. A variety of other nutrients mix well with polyphosphate fertilizers, making them excellent carriers of micronutrients typically needed by plants.
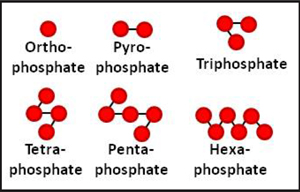
A single phosphate molecule is called an orthophosphate. When phosphoric acid and ammonia are reacted, water is driven off and orthophosphates begin to link together, chainlike, to form a “polyphosphate” fluid fertilizer. Each linkage of phosphate molecules has its own name deriving from its length, although polyphosphate is the general term that includes all of these linked molecules.
Agricultural use
In polyphosphate fertilizer, between half and three-quarters of the phosphorus is temporarily locked up by chained polymers. The remaining phosphorus (orthophosphate) is immediately available for plant uptake. With exposure to the soil, enzymes produced by soil microorganisms and plant roots break down the polymer phosphate chains into simple phosphate molecules the plants can digest, though some of the polyphosphates will decompose even without the enzymes. The enzyme activity goes faster in moist, warm soils. Typically, half of the polyphosphate compounds are converted to orthophosphate within a week or two for plant uptake. Under cool and dry conditions, the conversion may take longer.

Since polyphosphate fertilizers deliver nutrients both immediately and gradually via their combination of both orthophosphates and polyphosphates, plants can use them very effectively, longer.
Fluid fertilizers are commonly used in production agriculture, but not widely applied by homeowners. Farmers like them for their convenience and balanced nutrient distribution, since liquid fertilizers easily blend with many other nutrients and chemicals, and each drop of fluid is identical to the next. For most situations, growers choose between dry and liquid based on the price of nutrients, fertilizer-handling preferences and preferred field practices rather than significant agronomic differences.
Management practices
Most phosphorus-containing fluid fertilizers also contain ammonium, a substance primarily used as a source of P nutrition for plants. Since P has limited mobility in most soils, growers should strive to place the material as close to developing roots as practical. Best practices should be adopted to minimize the movement of P from the soil into adjacent water, since excess P in surface water can stimulate the growth of undesirable algae.
Non-agricultural use
Phosphate is an essential component in plant, therefore human, nutrition. Polyphosphate is an approved additive for food and requires no special handling precautions. Widely incorporated as a flame retardant on many products, including wood, paper, fabric and plastic, polyphosphate also functions as a commercial retardant for forest fires via the role its ammonium content plays in forming a protective charred layer after burning, thereby preventing further ignition.
Source: Nutrient Source Specifics (No. 2), International Plant Nutrition Institute.