In the mid-1970s, Dr. Robert Westerman banded 18-46-0 with wheat at planting in a low-pH soil near Haskell, Oklahoma. The impact was immediately evident. Soon after, Oklahoma State University published an extension brochure titled “Banding Phosphate in Wheat: A Temporary Alternative to Liming” (Figure 1). This method was a temporary solution for the significant amount of Oklahoma winter wheat that was either too far from a reliable lime source or under a short-term-lease contract.

Figure 1. The cover of an Extension brochure distributed in Oklahoma during the 1980s.
Today, grain producers throughout the United States commonly farm a large percentage of land that is not their own. Lease agreements can vary widely, both in length of term and the amount of inputs that the landowner will pay for. The wheat belt of Oklahoma is known for having large areas with low soil pH levels. A recent survey showed 38 percent of soil samples having a pH level below 5.5 (Figure 2). For farmers in Oklahoma, short-term leases with limited shared expenses have made remediation by lime applications impractical.Using lime in the dry environment of Oklahoma may result in a delay in correcting soil acidity by as much as one year. In a situation in which the lease agreement is only one to two years, there may be no economic benefit for the producer to apply lime, especially in regions where winter wheat average yields range from 20 to 40 bushels per acre.
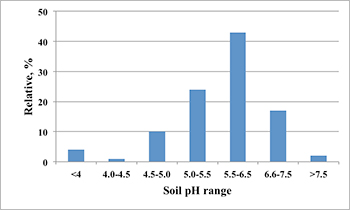
Figure 2. Summary of the soil pH values for the 614 samples submitted to the Oklahoma State University Soil, Water, Forage and Analytical Laboratory under wheat crop code during 2011.
Banding phosphorus is considered a bandage as the problem of soil acidity is not remediated, but simply addressed in the short term. Over time, the pH of the soil will continue to decrease. Where aluminum (Al) and manganese (Mn) toxicities are associated with soil acidity, phosphate reacts with those metals and creates a very insoluble solid compound. Phosphorus in high concentration in the seed zone reduces the harmful effects of the metals on the emerging seedling. Available Al, a predominant mineral in the region’s soils, is pH dependent. A change of 1.0 pH level changes available Al by 1,000-fold. For example, a soil with a pH of 5.0 will have an approximate Al concentration of 27 ppm (critical level toxic to winter wheat is 27 ppm), while a soil with a pH of 4.0 will have an Al concentration of approximately 27,000 ppm.
Aluminum and manganese toxicities not only impact grain yield, but also have an even greater impact on biomass production. A study published in 2007 showed that correcting soil acidity increased winter wheat grain yield by 82 percent and increased forage production by 150 percent (Kariuki, et al. 2007). In Oklahoma, wheat forage production is as important as grain, with approximately 50 percent of the 4 million acres of winter wheat in grazing. To maintain productivity on the land without the long-term investment of lime, farmers have been applying phosphorus fertilizer to alleviate the impact of aluminum toxicity. Research has also shown that the addition of banded phosphorus increases winter wheat forage production by two- to four-fold (Figure 3).
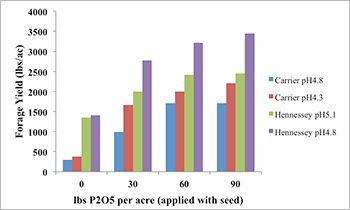
Figure 3. The impact of banding P fertilizer with seed at planting in four acidic soils on winter wheat forage production in Oklahoma. Chart adapted from Boman, et al., 1992.
While lime may take up to a year to change soil pH, banded phosporus has an immediate impact on the alleviation of metal toxicities. Figure 4 shows that the incorporation of lime improved forage yield but not to the degree of banding P. For continuous winter wheat producers, the time between application of lime and planting can be quite short. Typically, the previous crop will be harvested in mid-June, and sometimes, lime is applied as early as mid-July. At this point, there are only 60 days until the next wheat crop is planted in early to mid-September.
For the short-term tenant of a low-pH winter wheat field, the one-time investment of $60 to $120 per acre in slow-reacting lime is not an economically viable option. For a cost of approximately $25 per acre, winter wheat producers have used the in-furrow phosphorus fertilizer bandage for the past 40 years. Although not the ideal management practice, short-term lease agreements have driven wheat producers of the southern plains into alternative methods of nutrient application.
For more posts from Brian Arnall, follow his blog at http://osunpk.com/, or on twitter @OSU_NPK
Sources:
Boman, R.K., R.L. Westerman, G.V. Johnson and M.E. Jojola. 1992. Phosphorus fertilization effects on winter wheat production in acid soils. In Soil Fertility Highlights, Agronomy Department Oklahoma Agricultural Experiment Station, Oklahoma State University. Agronomy 92-1, 171–174.
Kaitibie, S., F.M. Epplin, E.G. Krenzer, Jr., and H. Zhang. 2002. Economics of lime and phosphorus application for dual-purpose wheat production in low-pH soils. Agron. J. 94:1139–1145.
Kariuki, S.K., H. Zhang, J.L. Schroder, et al. 2007. Hard red winter wheat cultivar responses to a pH and aluminum concentration gradient. Agron J. 99:88–98.